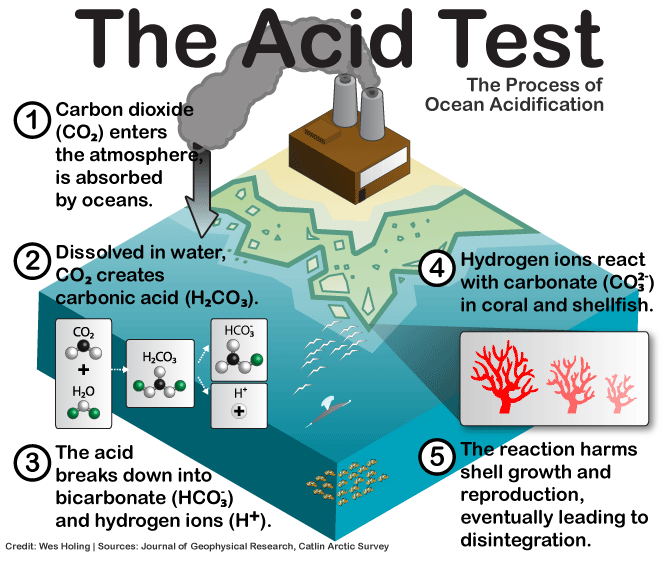
Once in the aqueous environment of the ocean, CO2 binds to water (H2O) to form carbonic acid (H2CO3) which quickly dissociates into a hydrogen ion (H+) and bicarbonate ion (HCO3-). Increases in hydrogen ion concentration are the definition of acidity, so this is where acidity increases, and that pesky carbon atom that we released form burning fossil fuels really starts to cause problems for marine life.
Normally, carbonate ions (CO3 2-) are used by marine life to build their shells, and healthy coral reef growth is directly related to carbonate concentration. Unfortunately, the excess H+ ions in the water combine with carbonate to make even more bicarbonate and reduce the carbonate concentration, ending in decreased shell growth, weak, malformed shells, and ultimately, significant degradation of coral reefs.
In the next post, we will look at how this process effects reefs and where in the world the impact is most significant.
Thanks for your interest!
 RSS Feed
RSS Feed